 Note that oxidation always happens at the anode and reduction always occurs at the cathode. (Remember: Anode is the electrode on which oxidation takes place, i.e., loss of electrons and cathode is the electrode on which reduction takes place i.e., gain. Also note that during oxidation, electrons are removed, so the oxidation number increases, and during reduction, electrons are added to the species, resulting in a decrease in the oxidation number. For charging the battery, the exact reverse of these reactions happens at the anode and cathode. Batteries Lead-Acid Battery The overall electrochemical reaction is PbO 2 (s) + Pb(s) + 2SO 4 2- (aq) + 4H + (aq) 2PbSO 4 (s) + 2H 2 O(l) for which E. Note: The above reactions occur during discharge of the lead storage battery, that is, when we are drawing current from it. The electrolyte in a lead storage battery is sulphuric acid in aqueous form: ion moves to the anode, gives up its two additional electrons becomes radical SO4, react with the lead sulphate anode and form leads peroxide and lead. We can adopt a similar strategy to find the reaction at cathode (where lead oxide would get reduced). As the battery charges, the sulfuric acid reacts with the lead in the anode and cathode to produce lead sulfate. The two electrodes are separated by an electrolyte of sulfuric acid. So, we can start off by writing the reactions of oxidation of lead with the help of the electrolyte and then balance it to get our equation. In a lead-acid battery, the cathode is made of lead-dioxide, and the anode is made of metallic lead. Oxidation happens at the anode and reduction happens at the cathode. 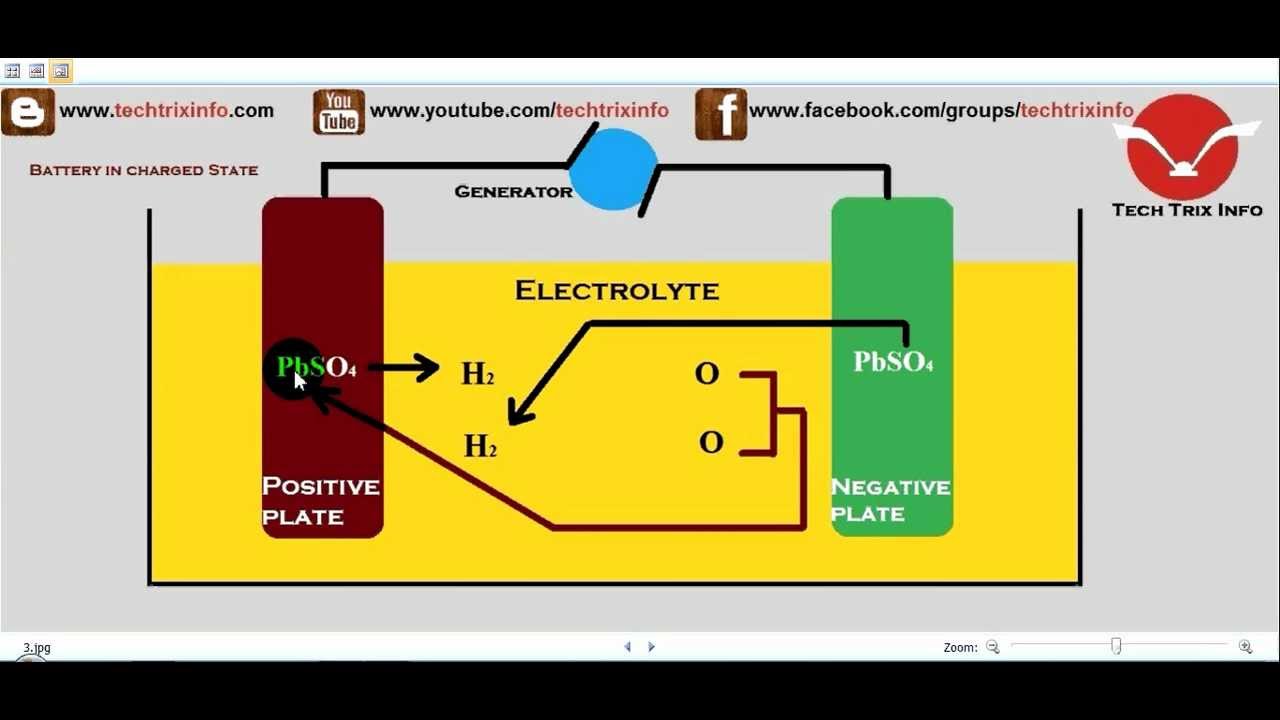 
Hint: In a lead storage battery, the cathode is lead dioxide and the anode is lead, while the electrolyte is an acid, usually sulphuric acid. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
